Challenging the Status Quo
Increase efficiency & quality in pharmaceutical production, powered by a modular validated platform for continuous manufacturing (MCM) .
What clients & partners say

“Through this project and our partnership with Zeton, we have provided an integrated continuous platform technology, which will allow a faster delivery of molecules to medicines to meet an ever greater patient need in a sustainable manner.”
Roger Shillitoe, AstraZeneca
How we push boundaries

Flexibility
Designed as standard building blocks, ContiUnity® adapts to your unique requirements, offering a perfect fit for numerous setups and processes.

Time to market
By selecting pre-validated ContiUnity® modules, you can bypass the design phase and configure your process plant quickly and efficiently.

Risk reduction
ContiUnity® is a multipurpose solution that can be quickly adapted to changing situations, eliminating the need for additional capital investments, resulting in cost reduction.

Sustainable
The intelligent modular design reduces floorspace by 70% compared to traditional batch facilities, with modules being reusable in future processes as well.

The new standard for the pharmaceutical industry
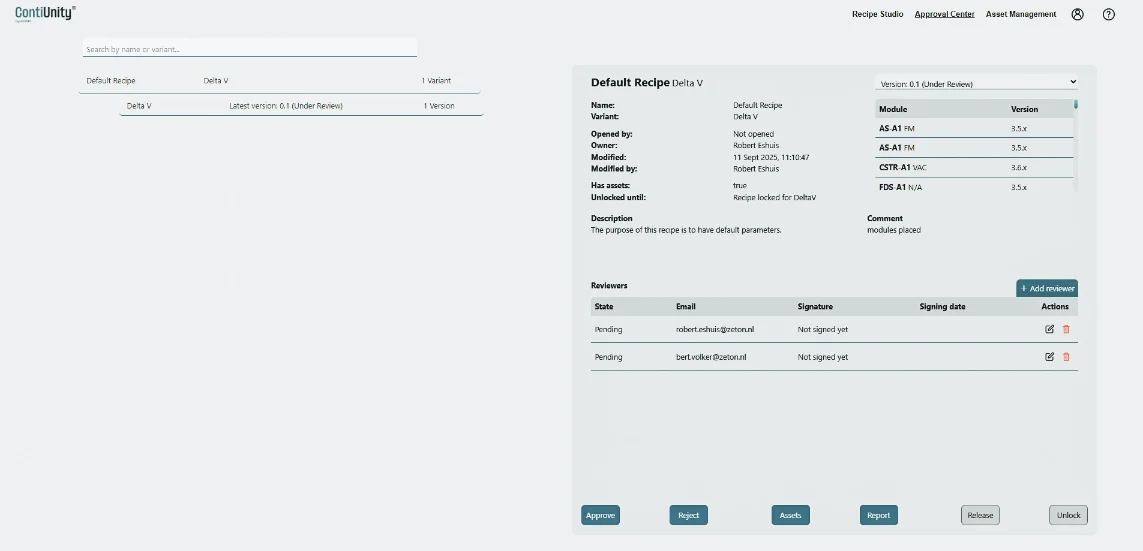
Developed in collaboration with leading pharmaceutical companies, ContiUnity® is the only pre-validated modular system for the continuous manufacturing of pharmaceutical active ingredients. It offers a complete solution that integrates modular hardware with intuitive process control strategies, tailored to your needs.
Project Approach
PROCESS TRANSLATION
Together with you, Zeton will translate your vision into the ContiUnity® architecture, tailored precisely to your needs.

MODULE SELECTION
Modules are selected from our library of pre-engineered units. If a custom module is required for a process, this can be designed in parallel.
ASSEMBLY & TESTING
At Zeton, we efficiently and skillfully assemble the complete system and subject it to rigorous IQ and OQ testing to ensure top quality.


PACKAGING & TRANSPORT
After successfully passing all tests, we will pack and ship the system. Limited disassembly is required due to ContiUnity®'s modularity.

ON SITE TESTING
With pre-validated modules and the system fully tested at Zeton, only limited Site Acceptance Testing is required before system handover.
















